Let’s Make Ethanol
Published
6/2/2014
Target Grade Level / Age Range:
9-12
Estimated Time:
2 class periods, plus one day for field trip
Purpose:
Students’ performance objectives are to 1) investigate the effects of temperature and enzymes in alcohol fermentation. 2) explore the process of ethanol production for use as biofuel, a renewable energy resource. 3) design and build an apparatus that measures gas production during fermentation.
Materials:
Essential Files
Vocabulary
- Alcohol: any compound that contains a carbon atom that is single-bonded to an oxygen bonded to a hydrogen atom (-OH group)
- Enzyme: a substance produced by a living organism that acts as a catalyst to increase the rate of a specific biochemical reaction.
- Ethanol: an alcohol containing only two carbons
- Fermentation: a normal metabolic process in which oxygen is not the final electron acceptor. Fermentation is the process through which alcohols are made and can occur in low oxygen conditions
- Distillation: a process of heating a liquid mixture to a specific temperature range in order to separate the substances by boiling point, then capturing and cooling the vapors.
- Fractional distillation: separation of a liquid mixture into its components (fractions) that differ in chemical composition (and hence boiling point) by means of distillation.
- Rate of reaction: rate at which reactants are used up or products are formed.
- Simple rate laws: A rate law is an expression that relates the reaction rate to the concentrations of the chemical species present in the reaction mixture, may include reactants, products and catalysts (but not reactive intermediates).
Background – Agricultural Connections
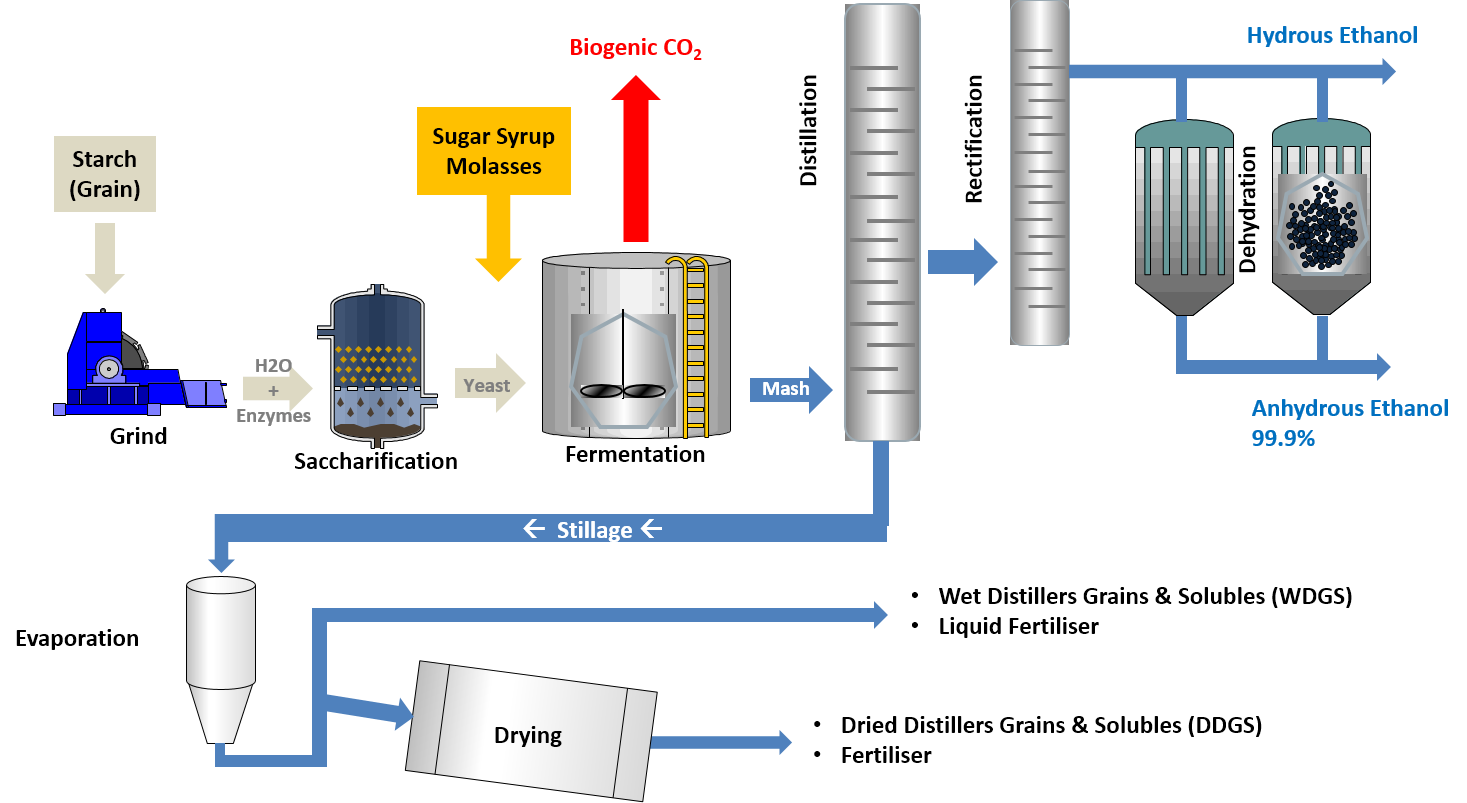
- Commercial production of fuel ethanol in the U.S. involves breaking down the starch present in corn into simple sugars (glucose), feeding these sugars to yeast (fermentation), and then recovering the main product (ethanol) and byproducts (e.g., animal feed).he chemical reaction of fermentation is where 1 mole of glucose yields 2 moles of ethanol and 2 moles of carbon dioxide. The reaction is:C6H12O6→2C2H6OH + 2 CO2
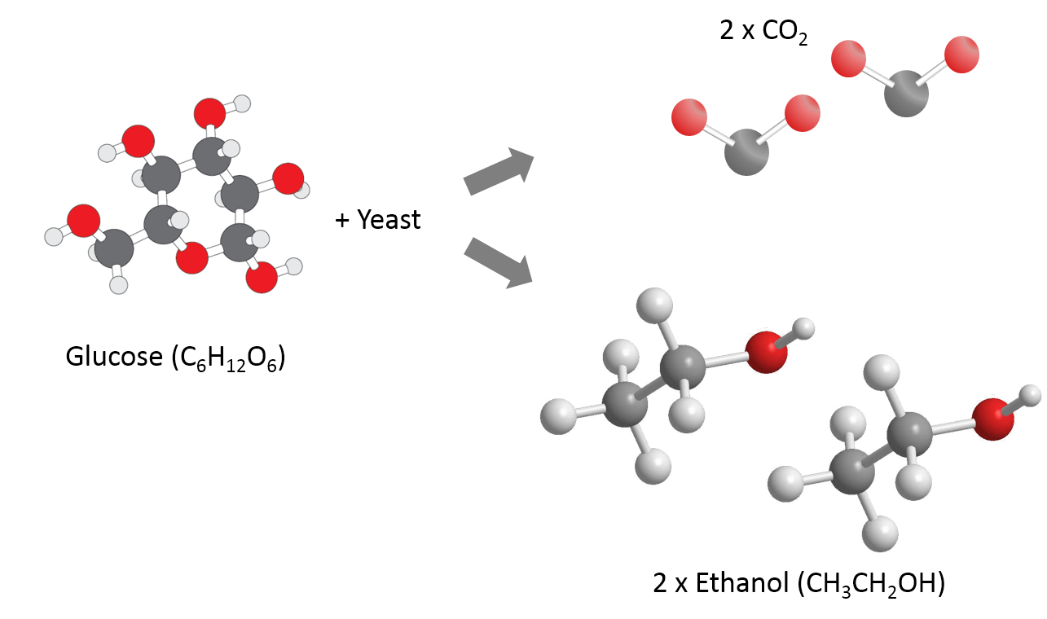
- The reaction takes place at 30-32°C for 2-3 days. Temperatures outside this range impact the fermentation process.
- Distiller’s grains is a coproduct of ethanol production. It is used as a source of protein and energy for livestock feed and contains the non-starch portions of the corn. It can be fed wet; or dried for ease of storage and transport. About one third of the weight of the corn used for the fermentation process remains as dried distiller’s grains (DDG).
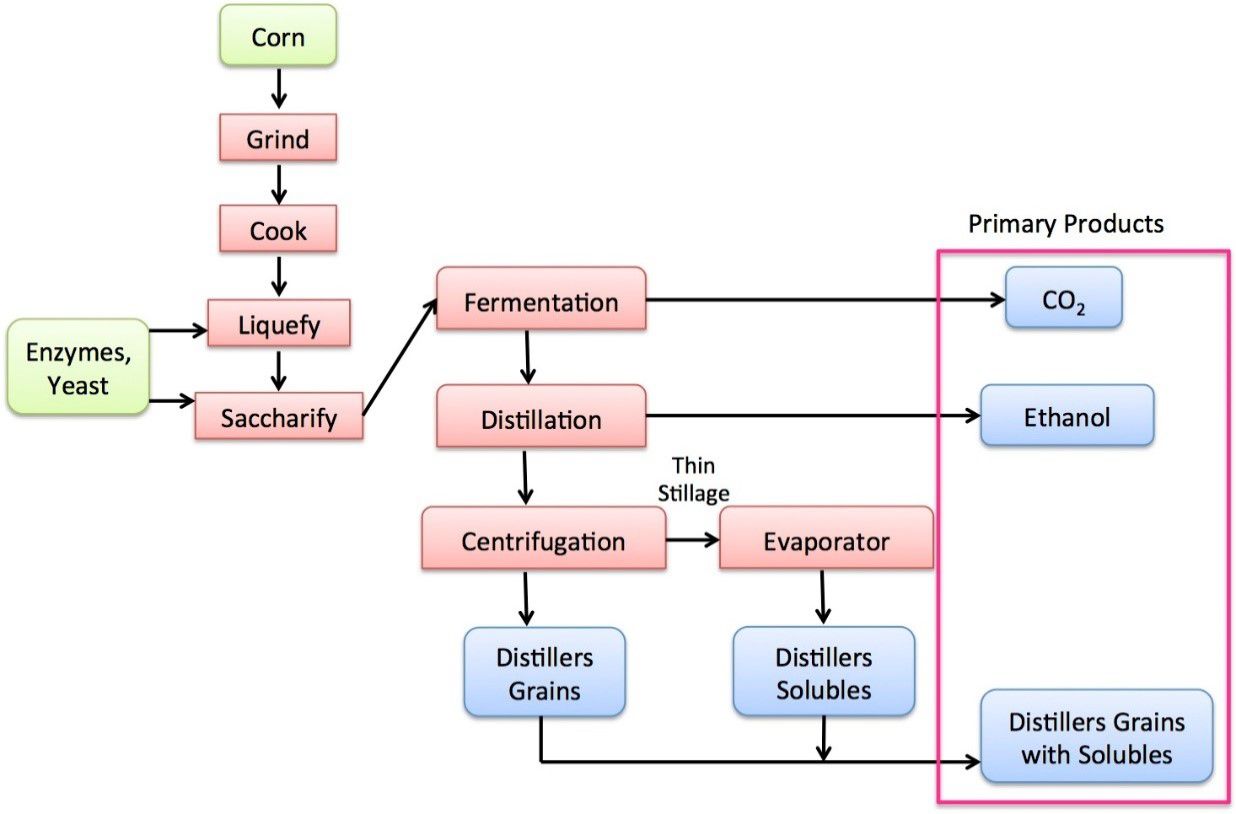
- Carbon dioxide produced during the fermentation of corn can be captured and used for beverages or chemical manufacturing.
- Like all living things, the yeast used in fermentation needs to have suitable living conditions in order to grow and metabolize. These factors include temperature, moisture, pH, and nutrients. The ethanol is produced as a waste of the yeast cells, concentrates in the liquid, and eventually kills the yeast and stops the fermentation process.
Interest Approach – Engagement
Students will make a visit, either in person or virtual via Farmchat, to an ethanol plant to learn about the chemical processes involved in the production of ethanol.
Procedures
- Day 1: Students will do research on the basic processes of ethanol production to develop a set of questions they will use during their visit to the ethanol plant. This research should include the basic chemistry behind fermentation and distillation. Guide student research with suggested websites, graphics from background information, and other sources as appropriate.
- Day 2: Students will make a real or virtual visit to the lab of an ethanol plant. They will use their background research to ask questions during the visit.
- Day 3: Students will produce their own ethanol (from sugar, not corn, to limit experimental variables). Using their knowledge of the ethanol production process, students will decide on the best method of production (considering temperature impact) and attempt to produce the most, high-quality ethanol. Use the Carolina STEM Challenge: Biofuels Kit Item #180950 and follow the instructions as provided. Making this a competition often encourages creativity and increased attention to details during the lab activity.
- Students will produce a lab report explaining their process and results.
Did you know? (Ag facts)
- Oil import dependence dropped to 41% in 2012 — the lowest since 1995. Without ethanol, oil import dependence would have been 48%.
- Ethanol reduces greenhouse gas emissions by 40-50% when compared directly to gasoline.
- In 2017, 14.6 billion bushels of corn were harvested in the United States with 2.6 billion produced in Iowa. Roughly 40% of US corn goes to ethanol production.
Extension Activities
- Check the percentages of ethanol in the fuels sold locally and compare that to other regions.
- Students can research ethanol production with other feedstocks (sugarcane, etc.) and in other nations.
- Students can set up experiments to test the effects of pH or other factors on fermentation rate.
- Ties to Social Studies-Use of ethanol in vehicle fuel is still quite controversial. Students could debate the pros and cons of regulation vs choice, economic impacts, effects on soil conservation, or other topics.
Suggested Companion Resources
- https://www.chemguide.co.uk/organicprops/alcohols/manufacture.html
- https://www.extension.purdue.edu/extmedia/id/id-328.pdf
- https://www.e-education.psu.edu/egee439/node/673
- http://www.worldofcorn.com/#corn-used-of-ethanol-and-ddg-production
Sources/Credits
- https://www.carolina.com/alternative-energies/carolina-stem-challenge-biofuels-kit/180950.pr
- http://www.westernfarmpress.com/management/40-interesting-facts-about-ethanol
- https://www.e-education.psu.edu/egee439/node/673
- http://biofuelsassociation.com.au/biofuels/ethanol/how-is-ethanol-made/
Author
Diane May
Organization Affiliation
West Delaware High School
Agriculture Literacy Outcomes
T4.9-12 g. Provide examples of how processing adds value to agricultural goods and fosters economic growth both locally and globally
Iowa Core Standards
- HS–PS1–5 Apply scientific principles and evidence to provide an explanation about the effects of changing the temperature or concentration of the reacting particles on the rate at which a reaction occurs.
- HS–ESS3–4 Evaluate or refine a technological solution that reduces impacts of human activities on natural systems.
- HS-ETS1-3. Evaluate a solution to a complex real-world problem based on prioritized criteria and trade-offs that account for a range of constraints, including cost, safety, reliability, and aesthetics, as well as possible social, cultural, and environmental impacts.
This work is licensed under a
Creative Commons Attribution 4.0 International License.
